Fda Prior Notice Login

Are food and food products.
Fda prior notice login. If you already have an account enter your username and password. Under the bioterrorism act of 2002 fda regulates foods entering the u s. Food and drug administration u s. In 2002 congress passed the bioterrorism act as a part of its.
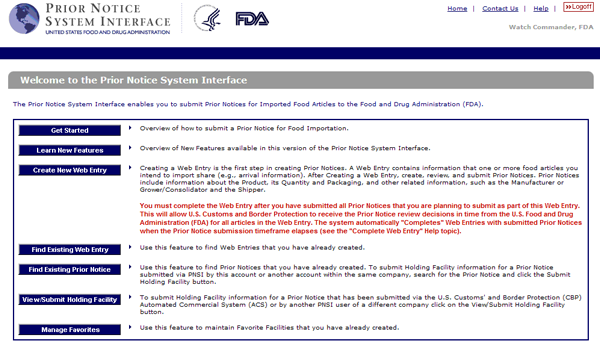
Fda requires the filing of prior notice for all food beverage and dietary supplements shipments entering the united states. Fis was created in part in response to the bioterrorism act of 2002 which gave high priority to improved information management to help protect the food supply the act requires that fda develop two systems. Food supply from outside the country through the prior notice of imported foods. Fda quickly and properly.
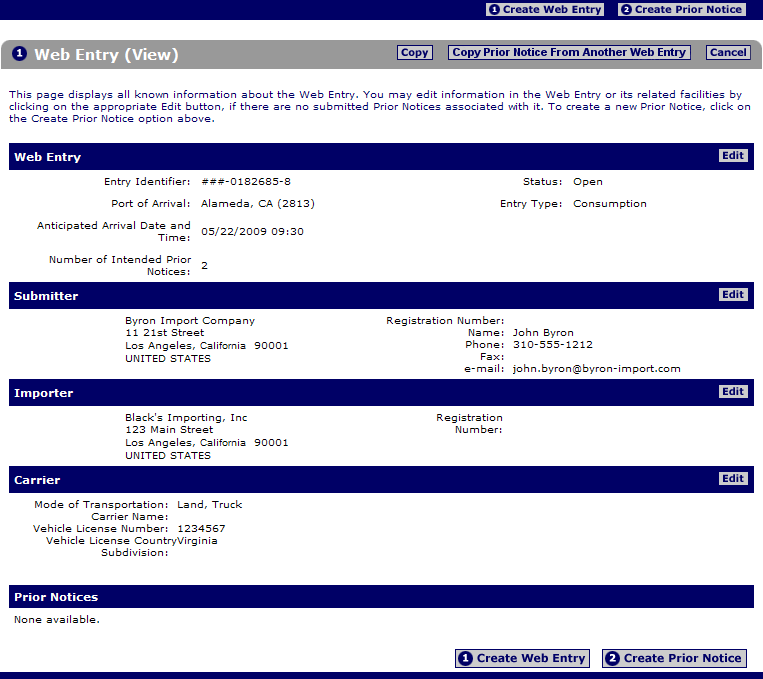
One to support the registration of facilities that manufacture process pack or hold food products intended for consumption in the united states and one to receive prior notice before. File prior notice with u s. Pn 0010 you are using an unsupported browser. Depending on the method of shipment express carrier air sea land rail mail or baggage fda may require different data elements to be submitted.
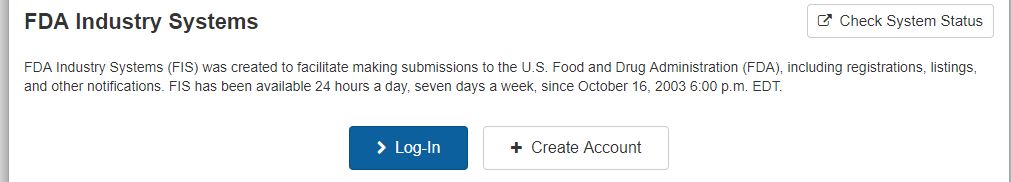
Introduction to prior notice requirements for imported foods. Nearly 20 of all imports into the u s. Upon receipt of prior notice fda will issue a confirmation number in the form of a bar code that must accompany most food shipments. To make submissions to fda e g establishment registration product listing and self id etc you must first create an account.
Food and drug administration u s. Fda online or via email or fax. Prior notice must be provided for all food for humans and animals that is imported or offered for import into the united states title 21 code of federal regulations 21 cfr part 1 subpart i. Fda requires prior notice for all foods imported into the united states.
Please refer to the computer system requirements for web browsers on the fda industry systems page www access fda gov. Fda requires the filing of prior notice for all food beverage and dietary supplements shipments entering the united states. Registrar corp s prior notice express allows you to quickly file prior notice with the u s.